Synthesis of mesoporous carbon aerogel by freeze drying method |
Quick View Leaflet
Carbon aerogels are ultra-lightweight, porous carbon materials with a 3D interconnected network structure, made by drying and then carbonizing organic gels. They have a high surface area, low density, and are electrically conductive, making them useful for applications like energy storage (supercapacitors, batteries), catalysts, and adsorption. Carbon aerogel is synthesized from resorcinol–formaldehyde gel in aqueous medium using a freeze-drying technique. The method involves gelation in a basic medium followed by curing at 75°C for 60h. The resulting aquagel obtained is frozen at –25 °C and sublimated under a vacuum of 10⁻²–10⁻³ bar for 4h, avoiding liquid–solid interface to prevent pore collapse. The dried organic aerogel is then pyrolyzed at 850°C in an inert atmosphere to obtain freeze-dried carbon aerogel (FD-CA). With the optimized synthesis parameters, carbon aerogel having total surface area of ~ 875 m2/g and mesopore surface area of >350 m2/g has been obtained. This process is standardized for bulk requirements and finds application as electrode material for supercapacitor, hybrid batteries and as a base matrix support for catalyst & fuel cells in various industrial and DAE applications
Detail Technical Brochure
Detailed Technical Brochure:
Carbon aerogel is a three-dimensional network of interconnected carbon particles having large specific surface area. It has unique features like tunable pore size, open meso-pore structure and good electrical conductivity.
- Synthesis Process:
Carbon aerogel is synthesized by gelation of resorcinol-formaldehyde with molar ratio 0.5, in aqueous medium using freeze drying technique. This process involves gelation, curing, drying and pyrolysis of organic gel to obtain carbon aerogel. Gelation is carried out in basic medium using sodium carbonate as catalytic agent with R/C ratio of 375. For complete gelation and strengthening of pore wall, gel is kept in oven for curing at 75 oC for 60 hrs to allow cross-liking of the network at steady phase. The aquagel obtained contains waterlocked in the network and is to be removed without disturbing the pores formed. For this, the aquagel is subjected to freeze-drying (FD) where gel is frozen below -20°C and then sublimation is carried out under vacuum of 10-2 to 10-3 bar for 6 hours avoiding formation of solid-liquid interface to prevent pore collapse. Pyrolysis of the dried organic aerogel is carried out at 850 oC in inert atmosphere to obtain carbon aerogel and is referred as FD-CA. Surface properties like BET surface area, mesopore area, pore volume, pore size distribution etc. of the FD-CA are evaluated by N2 gas adsorption technique-based surface area analyzer.
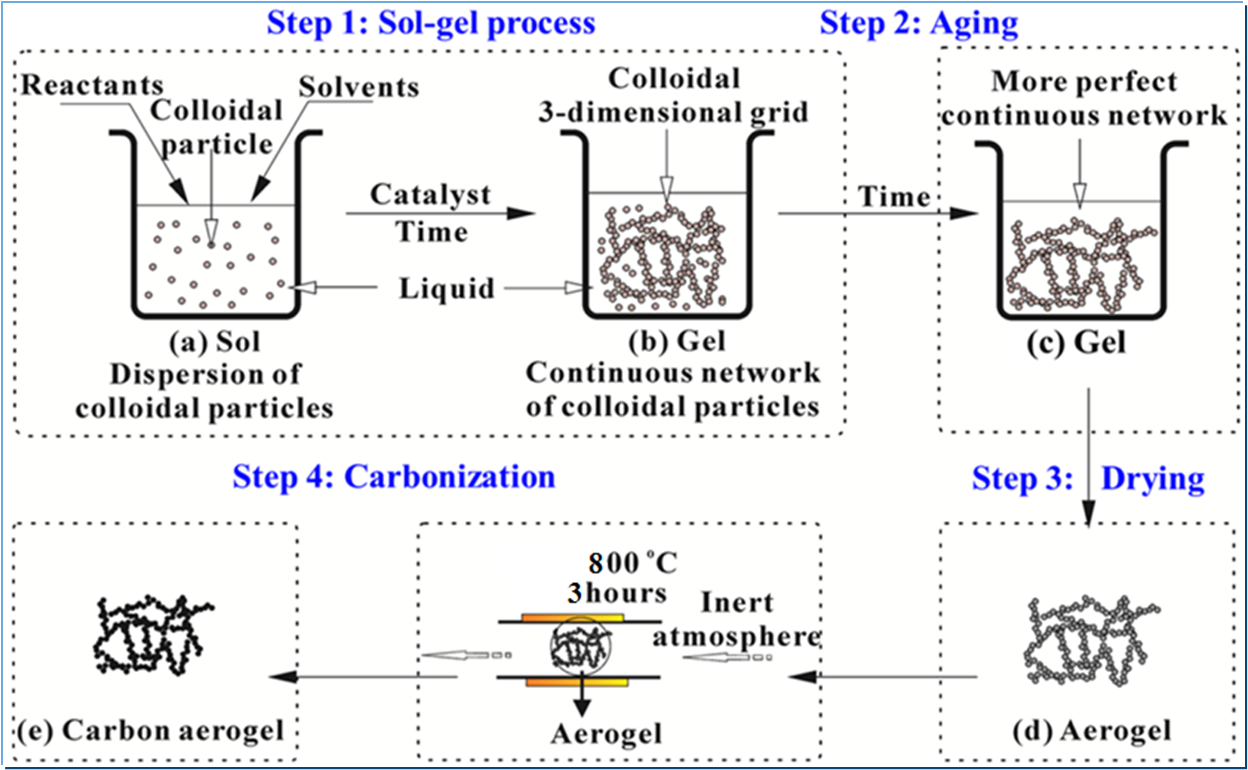 Figure 1. Steps involved in carbon aerogel synthesis
Figure 1. Steps involved in carbon aerogel synthesis
 Figure 2. Freeze drying setup using vacuum oven with moisture traps.
Figure 2. Freeze drying setup using vacuum oven with moisture traps.
- Technical Specification of developed Carbon Aerogel
|
S. No. |
Description |
Value |
|
a. |
BET Surface area |
~850 m2/g |
|
b. |
Mesopore area |
~350 m2/g |
|
c. |
Pore volume |
~1.1 cm3/g |
|
e. |
Average pore size |
~ 8 nm |
- Typical End Applications:
Carbon aerogels have diverse applications due to their large surface area, low density, high porosity and conducting nature. They are used as
- Energy storage:As electrodes for supercapacitors, lithium-ion batteries and fuel cells due to their high surface area and conductivity.
- Catalysis: Acting as a porous catalyst support in various chemical reactions.
- Adsorbents:Used for capturing substances like CO2 from the air.
- Desalination:For water purification processes. Electrodes for capacitive de-ionization of water
- Composite materials:When combined with polymers, they can significantly improve the polymer's electrical and mechanical properties.
- Applications specific to DAE:
- For electrode material for 100 V supercapacitor modules
- Support matrix for loading of precious metals (Pt, Pd etc.) for light element isotope exchange catalyst applications.
- Fuel cell electro catalyst supports
4. Facilities Required for Commercial Production
a. Floor Space (at least 50 sq m) required for synthesis & processing of freeze-dried CA
b. Ovens for curing, freeze drying setup and furnace for pyrolysis.
c. Utilities like argon gas, cooling water and electrical power.
5. Manpower Requirement
- One post-graduate (M Sc Chemistry) having at least two years’ experience in working similar field and
- one Technician (plant operator) with two years of experience in similar field.
